ODOR REMOVAL TREATMENT

1. Source of odor generation
In industrial production, odors can arise from a variety of sources:
- Odors from raw materials for processing of seafood, rubber, shrimp powder, chemicals, etc., of natural or artificial origin. The raw materials themselves have a specific smell due to the decomposition of organic compounds during transportation and storage.
- Odor from industrial manufacturing processes. During the manufacturing process, chemical odors, by-products or organic odors arise during the drying and distillation stages.
- Odor generated from the wastewater treatment system due to evaporation and biological fermentation in the area of the basins: collection, conditioning, grease separation, sedimentation, anaerobic treatment, aerobic treatment and sludge treatment.
- Odor pollutant components often contain groups of odorous substances that are easily quantified inorganic form such as ammonia, hydrosulphide, etc., or group of organic substances such as pesticides, organic solvents (methane, butane, benzene, xylene, cyclohexanone, toluene, etc.) or groups of substances that are difficult to quantify, evaporate at normal temperature conditions such as VOC, including many volatile organic substances, typically industrial chemicals in food manufacturing industry (spices), cosmetics, etc.,
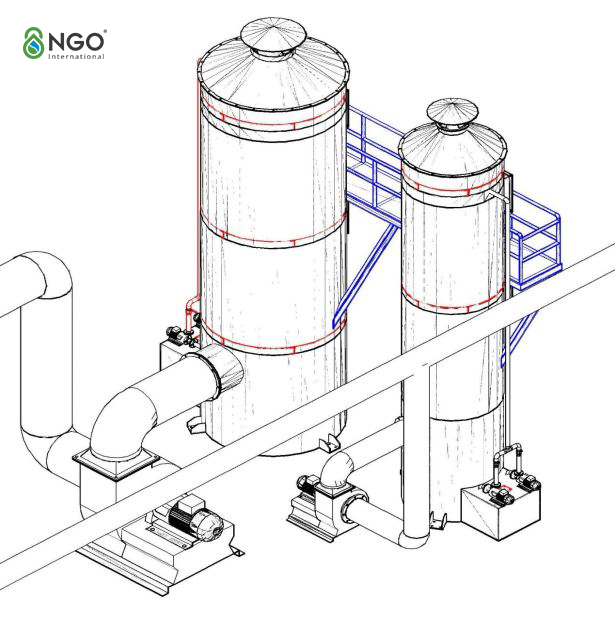
2. Some solutions to odor treatment
2.1. Absorption method:
2.1.1. Absorption by natricarbonate
- The exhaust gas mixture is fed into absorption tower 1 of the treatment system, which creates reaction H2S + Na2CO3 → NaHS + NaHCO3.
- The saturated solution from tower 1 will be directed to tower 2 and evaporated by hot air in the absorption tower to recover Na2CO3.
- Recovered Na2CO3 will be directed back to Tower 1 and repeat the cycle.
- In tower 2, the air drying process takes place and is blown by the 3rd fan to the top of the tower, leading to the next stage to recover the single sulphur and vanadate recirculation.
- Advantages: Easy to circulate and recover chemicals for the reaction process so it consumes less chemicals.
2.1.2. Treatment with NaOH
- Emission gas are sent to treatment tower 1. This is a buffer tower with a padded layer of Raschig porcelain.
- H2S gas is directed at the bottom of the tower and clean gas after absorption is released at the top of the tower. Absorbing solution is distributed evenly over the buffer and along the surface of the material. The absorption reaction takes place on the wet surface of the buffer material.
- The absorbed solution is put into the 2nd solution container.
- Part of the solution in container 2 is mixed with the new caustic soda solution in container 4 and the tower is returned to the tower with pump 6 to continue the new cycle.
- The remainder of solution 2 is passed through a lime chloride treatment container (container 3) before being discharged into the environment.
- The concentration of alkali in the absorbing solution is controlled at 7g /l.
- Gas velocity in the tower is 0.6 m / s.
- The amount of NaOH consumed is converted to 8 kg of sulphur.
- Can replace NaOH with CaCO3.
- Advantages: Ensure 100% H2S is treated in exhaust gas; Treatment system does not require made of acid resistant material; Gas washing equipment with padding is also capable of lowering the temperature and filtering wet dust in the exhaust.
- Disadvantages:
Difficulties in cleaning buffer materials.
Easy to clog buffer materials due to the process of sediment accumulation.
2.1.3. Treatment with NH3
- H2S gas is fed into the bottom of the pyramid absorption tower, NH3 absorption solution is flowed from the top down through the top of the pyramid, in phase contact with the upward gas flow, where absorption occurs. Clean air is taken outside the tower.
- The solution is absorbed through container 2 and distillation tower No. 6, (NH4) 2S is broken down into NH3 and H2S at appropriate temperature and pressure environment. At the tower, steam distillation takes place to separate H2S from the solution and bring it out, the remaining NH3 is passed through the cooling equipment before re-circulating the absorption tower.
- H2S continues through the process of acid preparation, or single sulphur.
- Advantages:
100% absorption solution during the process of recirculation
Simple process, widely applied.
Materials are easy to find, cheap prices.
- Disadvantage:
It takes time to cool down the desorption process.
Bad smell
2.2. Adsorption method
2.2.1. Adsorbed by iron oxide
- For simple parallel filter systems:
Including many filters mounted in parallel to the direction of the gas, each vessel has several layers of adsorbent to pass gas layer after layer. Each layer of material is about 0.3 - 0.5 m thick. The air valve system on the pipe allows cutting any saturated absorption vessel to revert. If the adsorption vessel has 4 layers, the efficiency is 99-99.9%.
Emissions containing H2S will follow the air inlet system into the filter, then retained in the filter material by reaction: Fe2O3 + 3H2S → Fe2S3 + 3H2O. Then clean air is taken out.
The reaction occurs until the layer of filter material is saturated, the air system will disconnect to revert according to the reaction: 2Fe2S3 + 3O2 → 2Fe2O3 + 6S. The filter media layer will be replaced and further processed.
The adsorbent layer is considered to have no effect when S accounts for 50% of the material.
- For absorption tower system: Simple operation as simple filter, but when reconstituted will blow oxygen together to treat the reconstituted in parallel.
- Advantages:
Simple process, easy to implement.
Very high performance,> 90%.
Cheap materials.
- Disadvantages: It takes time to change the filter media
2.2.2. Adsorption by activated carbon
- The air needs to be filtered by an air supply fan blowing into adsorption tower 2, where H2S oxidation occurs on the activated carbon surface.
- The sulphur released in the reaction gradually accumulates on the surface of activated carbon. The treated gas is passed through Cyclone 1 to filter dust and adsorbent and passes through the subsequent processing stages.
- After the adsorbent becomes saturated with S, the adsorbent is carried out with (NH4) 2S which is put into decomposition tower 7 to obtain a single S.
- Sulphur is collected by the difference in unit mass, while condensation is slightly condensed during (NH4) 2S.
- After separating S from saturated coal, people wash coal with water until there is no more SO2 in the water. The charcoal is then dried for reuse.
- Coal is transported by lift bucket 6, to conveyor drying house 5, through hopper 3 to put into adsorption tower 2 to continue the process. The adjustment of coal flow before entering the tower thanks to the flow control device 4.
- Advantages:
Simple process, easy to implement
High efficiency,> 90%.
High purity of sulphur: 99.9%
- Disadvantages: Exhaust gas needs to filter dust before putting into the adsorption system (dust concentration of 2-3 mg/m3)
If your business is interested in solution to inlet water treatment, or has a demand for ion exchange materials, please contact NGO via phone number (024) 3566 8225 or email office@8ngo.com for direct consultation.
-
 Wastewater treatment
Wastewater treatment
-
 A standard domestic wastewater
A standard domestic wastewater
-
 B standard domestic wastewater
B standard domestic wastewater
-
 Pig Farm Wastewater
Pig Farm Wastewater
-
 Textile dyeing wastewater
Textile dyeing wastewater
-
 Paper industry wastewater solution
Paper industry wastewater solution
-
 Textile Dyeing Wastewater & Industrial Washing
Textile Dyeing Wastewater & Industrial Washing
-
 Slaughter wastewater treatment solution
Slaughter wastewater treatment solution
-
 Aquaculture wastewater
Aquaculture wastewater
-
 Starch wastewater
Starch wastewater
-
 Petroleum wastewater
Petroleum wastewater
-
 Beer wastewater
Beer wastewater
-
 Other industrial wastewaters
Other industrial wastewaters
-
 BioPM - Organic Industrial wastewater treatment solution
BioPM - Organic Industrial wastewater treatment solution
-
 MBR solutions
MBR solutions
-
 Conventional activated sludge (CAS)
Conventional activated sludge (CAS)
-
 Moving bed biofilm reactor (MBBR)
Moving bed biofilm reactor (MBBR)
-
 Anaerobic- Anoxic-Aerobic (AAO)
Anaerobic- Anoxic-Aerobic (AAO)
-
 Sequencing batch reactor (SBR)
Sequencing batch reactor (SBR)
-
 Physical & Chemical methods
Physical & Chemical methods
-
 Supply water treatment
Supply water treatment
-
 Cooling system water
Cooling system water
-
 Water treatment for food and Beverage production
Water treatment for food and Beverage production
-
 Ultra pure water supply solution for electronic industry
Ultra pure water supply solution for electronic industry
-
 Pure water supply for pharmaceutical manufacturers
Pure water supply for pharmaceutical manufacturers
-
 Water Supply treatment with ion exchange method
Water Supply treatment with ion exchange method
-
 Ordor Treatment
Ordor Treatment

