HIGH-TECH WASTEWATER TREATMENT SOLUTIONS IN THE PLATING INDUSTRY
1. Discharge source
1.1 Classification of plating technology:
-
Electroplating: is the most widely used method today. Electroplating is the process of electrochemical cathode poles. Metal surface as cathode in an electrolytic vessel using an external DC power source, the plating solution is anode containing metal ions to be plated. These metal ions participate in cathode reaction and are de-electrochemical into electrolytic metal and precipitate on the surface to be treated.
-
Chemical plating: is a method based on chemical reduction, metal ions are reduced to metal from its salt solution by reducing agents. The electronics required for the reduction process are provided by chemical reducing agents. For example, in Nickel Plating, sodium hypophosphite is used as a reducing agent
-
Hot dip galvanizing: is the process in which the material to be plated passes through a metal storage tank (pure metal), which is melted at a high temperature, resulting in the metal plating on the surface of the materials to be plated.
1.2 Basic plating process:
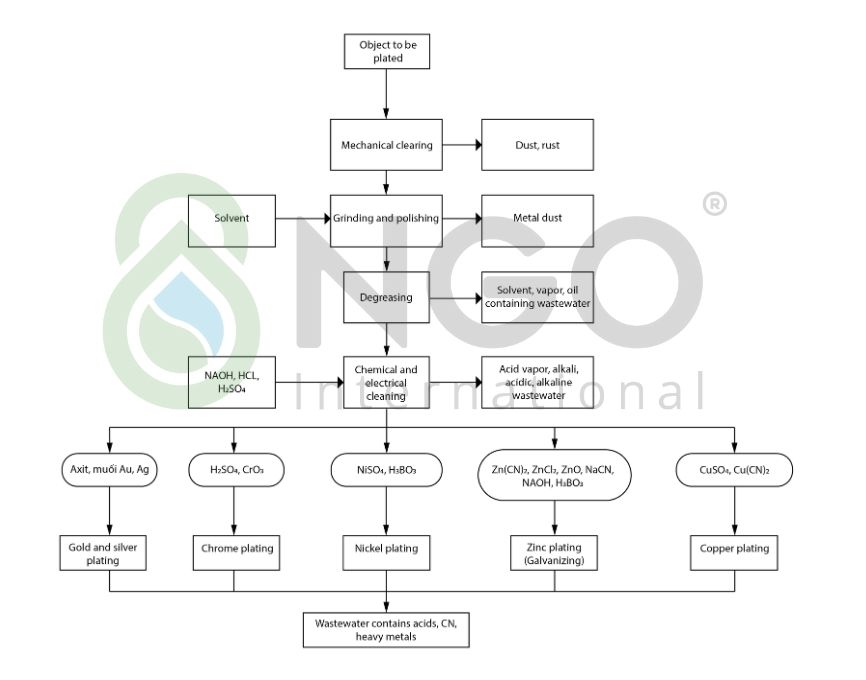
1.3 Sources of wastewater:
-
Wastewater from the plating process: The solution in the plating tank can leak, scatter or stick to the plating jigs and outward details. After a period of operation, the plating tanks need to be cleaned by discharging dirt and sediment along the wastewater stream. Therefore, the amount of wastewater is not much, but pollutants are diverse, with a high concentration of pollutants (Cr6 +, Ni2 +, CN–…)
-
Wastewater from surface cleaning process: There is grease on metal surfaces due to mechanical curing and polishing. To ensure the coating quality, the parts need to be cleaned by chemical degreasing, solvents or electrochemical before plating. Therefore, the volume of wastewater generated in this process is large but the concentration of pollutants is low, mainly containing alkalis, acids and solutions.
2. Composition and characteristics of wastewater
-
Wastewater from plating process contains a lot of heavy metals, which has significant long-term impact on the environment, leading to future consequences because if the wastewater is not treated properly, metal ions, salts and other compounds will exist and be accumulated in the natural environment instead of being degraded when they are released into the environment.
-
This type of wastewater can destroy plankton, cause poisoning and disease to aquatic animals, especially fish (one of the links in the food chain in which humans are involved) that causes dangerous diseases in humans. The process is called bioaccumulation in which toxins derived from metals are released into the environment, and gradually accumulate in aquatic animals for a long time, following many food chains, affecting to people. Therefore, it is necessary to have a plating wastewater treatment system that separates metal ions before being discharged into the environment in accordance with the prescribed standards in Vietnam.
-
Plating wastewater has a variety of concentrations, pH varies widely from being acidic (pH = 2 - 3) to very alkaline (pH = 10 - 11). A common feature of plating wastewater is that it contains a high content of inorganic salts and heavy metals. Depending on the metal of the coating, the source of pollution can be Cu, Zn, Cr, Ni, etc., and depending on the metal salt used, the wastewater contains toxins such as cyanide, sulphate, ammonium, chromate, etc. Organic substances are rarely found in plating wastewater, the main part is flocculants, surfactants, etc. Therefore, the BOD, COD or grease are often low and not main objects of the treatment process. The main treatment objects are inorganic ions, especially heavy metal salts such as Cr, Ni, Cu, Fe, Zn, etc., with high pH and high temperature.
-
For factories using industrial salt in the production process, wastewater often has high nutritional Ammonium and Nitrogen content, even high chlorine generated by the salt electrolysis process, which are quite difficult to be treated and the cost of treatment is high.
-
Typical plating wastewater characteristics (*) Pollution values do not include high-concentrated pollution effluents
3. Popular process for plating wastewater treatment in Vietnam
Current wastewater treatment system applies a chemical precipitation method for metals present in the wastewater. This method should strictly follow reaction conditions for maximum removal of heavy metals. The typical metals of the wastewater stream are: Chromium, Zinc, and Nickel are all amphoteric metals. Their chemical precipitates can all dissolve in the excess alkaline environment. Thus, if the pH control process is not followed, the metal will not be removed as much as possible so that the water quality can be improved to meet QCVN 40: 2011/BTNMT.
The chemicals commonly used to treat plating wastewater are FeSO4, FeCl, H2SO4, Na2S2O5, PAC, Polymer.
-logo.png)
4. NGO's solutions to wastewater treatment
LESS OR NO CHEMICAL USED to remove heavy metals and other pollutants in water, which helps reduce chemical costs, with easier and less toxic operation, reducing sludge generated, limit the cause of secondary pollution.
Because of chemical-free and not subject to PH conditions to coagulate each metal component, the NGO's solution allows to treat all wastewater effluents generated in the plating process with no separation of currents, thereby minimize number of tanks and the complexity of treatment process, and simplify the wastewater collection systems from various production segments.
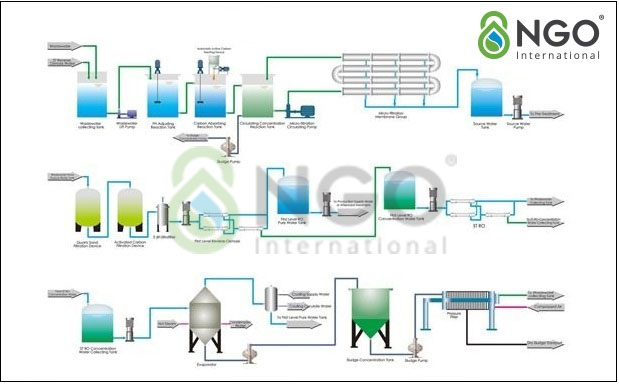
4.1 Plating wastewater treatment solution using various types of filtration technology.
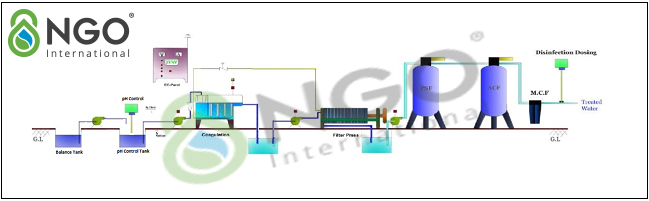
4.2 Plating wastewater treatment solution using electrochemical technology.
4.3 Water sample after treatment

If your business is interested in solution to inlet water treatment, or has a demand for ion exchange materials, please contact NGO via phone number (024) 3566 8225 or email office@8ngo.com for direct consultation.
thong_tin_lien_quan
-
 Wastewater treatment
Wastewater treatment
-
 A standard domestic wastewater
A standard domestic wastewater
-
 B standard domestic wastewater
B standard domestic wastewater
-
 Pig Farm Wastewater
Pig Farm Wastewater
-
 Textile dyeing wastewater
Textile dyeing wastewater
-
 Paper industry wastewater solution
Paper industry wastewater solution
-
 Textile Dyeing Wastewater & Industrial Washing
Textile Dyeing Wastewater & Industrial Washing
-
 Slaughter wastewater treatment solution
Slaughter wastewater treatment solution
-
 Aquaculture wastewater
Aquaculture wastewater
-
 Starch wastewater
Starch wastewater
-
 Petroleum wastewater
Petroleum wastewater
-
 Beer wastewater
Beer wastewater
-
 Other industrial wastewaters
Other industrial wastewaters
-
 BioPM - Organic Industrial wastewater treatment solution
BioPM - Organic Industrial wastewater treatment solution
-
 MBR solutions
MBR solutions
-
 Conventional activated sludge (CAS)
Conventional activated sludge (CAS)
-
 Moving bed biofilm reactor (MBBR)
Moving bed biofilm reactor (MBBR)
-
 Anaerobic- Anoxic-Aerobic (AAO)
Anaerobic- Anoxic-Aerobic (AAO)
-
 Sequencing batch reactor (SBR)
Sequencing batch reactor (SBR)
-
 Physical & Chemical methods
Physical & Chemical methods
-
 Supply water treatment
Supply water treatment
-
 Cooling system water
Cooling system water
-
 Water treatment for food and Beverage production
Water treatment for food and Beverage production
-
 Ultra pure water supply solution for electronic industry
Ultra pure water supply solution for electronic industry
-
 Pure water supply for pharmaceutical manufacturers
Pure water supply for pharmaceutical manufacturers
-
 Water Supply treatment with ion exchange method
Water Supply treatment with ion exchange method
-
 Ordor Treatment
Ordor Treatment


_-_NGO__2_jfif_da_sua.jpg)

